The National Haemophilia Database
The NHD collects, holds, processes and analyses confidential data for both research (non-invasive / non-interventional ) and non-research purposes (direct care, planning and commissioning). This information is kept in the National Haemophilia Database, which is located in Manchester. You need to be on the secure NHS network to access the database.
The National Haemophilia Database is being redeveloped to make it not only a fit for purpose database for the modern digital age but will enable us to develop new services and provisions for all key stakeholders that will take us to 2030 and beyond. Roche, CSL Behring, Pfizer, Sobi and Takeda has provided a financial grant to support the redevelopment of the National Haemophilia Database.
2021 Information Governance Assessment of the National Haemophilia Database – We are compliant!
The Information Governance Team at Manchester University NHS Foundation Trust (MFT) conducted an inspection of the NHD office and additionally, all our information governance arrangements including our polices, processes and procedures to assess our compliance with the Data Protection Act 2018 (DPA); General Data Protection Regulation (2016); Caldicott Principles and prevailing NHS Information Governance, guidance, legislation, and good practice as appropriate.
The inspection concluded on 30 June 2021, where MFT provided a comprehensive report on our arrangements which covered the following inspected areas: the NHD’s purpose, governance, staffing, suppliers, transparency, subject access requests, data quality and technical controls.
MFT’s report provides assurance that all our arrangements are compliant with all the relevant information governance regulations, Caldicott Principles and NHS legislation, guidance, and best practice. Their summary of the outcome can be found below:
MFT Summary:
“The Information Governance framework surrounding the National Haemophilia Database demonstrates a high level of process maturity. The governance actively engages data subjects in decision making about how their information is used and the management team were able to evidence this. There is appropriate separation of concerns in the design of the organisational structures with the intention of protecting personal information. As a small organisation they have engaged with suppliers who have the necessary credentials to provide a secure IT solution. The organisation is about to embark on a major upgrade of its technical platform. A further audit will be appropriate once this work has been completed.”
Patient Information and Consent Changes
Change of research registry name
Following consultation with patients and the Haemophilia Society, retaining the research registry for haemophilia would not be inclusive of all individuals who have different bleeding disorders to that of haemophilia. Based on this feedback, we believe it is the right time to change the name of the research registry. We have therefore changed it to the UK Bleeding Disorders Research Registry (previously known as UK National Haemophilia Research Registry).
Renewal of research registry ethical approval
In November 2025, we were successful in applying the Health Research Authority Research Ethics Committee (REC) to renew our ethical approval to undertake research. The UK Bleeding Disorders Research Registry (previously known as UK National Haemophilia Research Registry) has had its ethical approval to undertake research renewed for a further 5 years until 21 November 2029 (IRAS Project ID 339857). UKHCDO is the data controller and custodian for the data in both the National Haemophilia Database and for the UK Bleeding Disorders Research Registry (UK-BDRR).
Legal basis – all UK regions
As part of our application for renewal, the HRA agreed that for England and Wales, we could have a dual legal basis for collecting, holding, processing and analysing individuals’ data for research purposes.
We agreed a strategic roadmap to roll out a new explicit consent process for all Haemophilia Centres across all UK regions (England, Wales, Scotland and Northern Ireland) to undertake with all individuals with bleeding disorders who are in receipt of treatment at their centre. This has now been implemented and will continue for at least the next five years.
In December 2025, we were successful in applying to the Health Research Authority Confidentiality Advisory Group for Section 251 support in England and Wales. This in effect permits the NHD to collect, hold, process and analyse confidential data for both research and non-research purposes and to transfer confidential data between parties without there being a breach of our common law duty of confidentiality. We applied for this for the following reasons:
- If an individual with a bleeding disorder is lost to follow up, or
- If any individual with a bleeding disorder recorded in the database dies, we can be notified of their deaths and the details such as the date and cause, without obtaining informed consent. It is an NHS Digital requirement that we have this support when applying for mortality data.
For Scotland, in August 2025 the Scottish Public Benefit and Privacy Panel for Health and Social Care (PBPP) gave approval for Haemophilia Centres in Scotland to roll out the new consent process to use as the legal basis to share data with the NHD to enable it to collect, store and process confidential data for research and non-research purposes. In September 2021, PBPP also gave permission for processing data without written consent from the individuals concerned where there are practical difficulties in obtaining written informed consent, for example, from people lost to follow up or deceased. This dual legal basis will continue in Scotland.
For Northern Ireland, consent will continue to be obtained from patients. We have a data sharing agreement in place, which is reviewed and renewed by the Strategic Planning and Performance Group (SPPG, formerly known as the Health and Social Care Board), which is part of Health and Social Care Northern Ireland.
We have drafted new patient information which have been consulted on with the Health Research Authority, NHS Digital, NHS Scotland, Northern Ireland Privacy Advisory Committee, HSC Business Services Organisation (NI), UKHCDO members and patient representatives. During this process we have simplified the amount of information where it has been possible to do so.
The following leaflets cover all England, Scotland and Wales regions only.
UK Bleeding Disorder Adult Information Leaflet and Consent Form – This is the digital version 2.0 June 2025 leaflet that can be used in Haemophilia centres for all England, Scotland and Wales.
UK Bleeding Disorder Child (6-10 years) Information Leaflet and Assent – This is the digital version 2.0 September 2024 leaflet that can be used in Haemophilia centres for England, Scotland and Wales.
UK Bleeding Disorder Young People (11-15 years) Information Leaflet and Assent – This is the digital version 2.0 September 2024 leaflet that can be used in Haemophilia centres for England, Scotland and Wales.
UK Bleeding Disorder Young People (11-15 years) Information Leaflet and Consent Form for Those Able to Give Consent – This is the digital version 2.0 September 2024 leaflet which can be used in Haemophilia centres for England, Scotland and Wales.
UK Bleeding Disorder Parent/Guardian Information Leaflet and Consent Form – This is the digital version 2.0 September 2024 leaflet that can be used in Haemophilia centres for England, Scotland and Wales.
Data Opt-Out Form – This form covers all UK regions (England, Wales, Scotland and Northern Ireland). We need to make sure we are explicit in providing a process for patients to opt-out of their data being used for research purposes. The opt-out form will be made available to all Haemophilia Centres. Patients will also be able to contact the NHD directly.
The following leaflets cover the Northern Ireland region only.
UK Bleeding Disorder Adult Information Leaflet and Consent Form – This is the digital version 2.0 June 2025 consent form that can be used in Haemophilia centres for Northern Ireland.
UK Bleeding Disorder Child (6-10 years) Information Leaflet and Assent – This is the digital version 4.0 July 2025 leaflet that can be used in Haemophilia centres for Northern Ireland.
UK Bleeding Disorder Young People (11-15 years) Information Leaflet and Assent – This is the digital version 3.0 July 2025 leaflet that can be used in Haemophilia centres for Northern Ireland.
UK Bleeding Disorder Young People (11-15 years) Information Leaflet and Consent Form for Those Able to Give Consent – This is the digital version 3.0 July 2025 leaflet which can be used in Haemophilia centres for Northern Ireland.
UK Bleeding Disorder Parent/Guardian Information Leaflet and Consent Form – This is the digital version 3.0 July 2025 leaflet that can be used in Haemophilia centres for Northern Ireland.
Patient Consent form – Haemophilia Centres staff should use the NHD website (https://nww.mdsas.nhs.uk/nhd) to access patient consent forms. This requires access to the secure NHS network and a valid user account.
Access to National Haemophilia Database health records application form
Please download and complete the Subject Access Request form to request you Health Records from the National Haemophilia Database. Included in this document are all the necessary details of how to do this. If you are a patient representative please download the form specifically for a Patient Representative.
National Haemophilia Database Dataset
This dataset is consulted on with key stake holders for direct patient care, planning, commissioning, and research purposes through the Data Management Working Party
This includes lists of additional data points collected if an adverse event, such as a new factor VIII inhibitor, is reported to the database.
This list bears no relationship to data collected in previous years and should not be taken as a guide to the information collected in the past. Data collected in the earlier years of the database was very limited but has widened gradually with the passage of time.
Download the NHD Dataset 2020 –  650kb
650kb
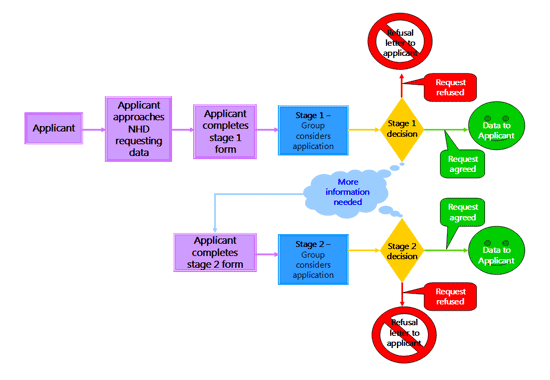
Obtaining Data from the National Haemophilia Database
To request data from the the National Haemophilia Database, complete the form.
The procedure for reviewing an application can be viewed below.
Please read all of the questions prior to completing the form, they can be found here.
Request for data from the NHD flow
NHD Contact Details
| Administrators: |
|---|
| Mr Greg Shone UKHCDO Suite 1, 2nd Floor of Anchorage One Anchorage Quay Salford Quays M50 3YJ |
| Tel : 0161 850 8102 Email: support@ukhcdo.org |